This week, we look at what continuous glucose monitoring (CGM) systems, insulin pumps and other tools have in store for us.
Continuous glucose monitoring systems

The Freestyle Libre 3 will automatically send blood sugar results to a smart phone, eliminating the need to scan the sensor. The sensor will also be smaller and thinner than the current model.

The Dexcom G7 will combine the sensor (which is inserted into the skin) and the transmitter (which is installed on the sensor and sends data to a Dexcom receiver or smart phone) into a single, smaller, disposable device that will last longer (about 14–15 days).
Insulin pumps
Tandem T-Sport and a new algorithm for the t:slim X2

The Tandem T-Sport is a mini insulin pump about half the size of the current Tandem pump.
The tubing is a lot smaller, and the pump doesn’t have a screen and is controlled with a mobile app. The U.S. launch was planned for early 2021.
Tandem is also about to launch a mobile app in the U.S. to control the t:slim pump remotely and a new algorithm with more precise settings, which will eventually replace the Control-IQ technology, available in Quebec since March 2021 (which also replaces the Basal-IQ technology).
We can usually expect these new products to be available 18 to 36 months later in Canada.
Omnipod Horizon

The highly anticipated Omnipod Horizon will be Insulet’s first hybrid closed loop system (artificial pancreas). The Omnipod pump works with a pod; it is the only tubing-free infusion system. The new Horizon system will be designed to be paired with the Dexcom CGM, but should also work with the Freestyle Libre.
Users will be able to set their glycemic target range between 6.1 and 8.3 mmol/L and control their pump with their phone.
The company has not yet announced whether it will keep manufacturing the current pods, which are used by some patients as part of their DIY artificial pancreas or looping system.
Medtronic 780G and MiniMed Duo

MiniMed 780G
This new hybrid closed loop system will follow the 770G pump, available in Quebec since March 2021.
The algorithm will calculate correction boluses that will be given automatically (as the current Tandem t:slim X2 does) to correct hyperglycemia, and the user will be able to set a glycemic target between 5.5 and 6.7 mmol/L. It will be paired with the Guardian 3 CGM as well as the next Medtronic CGM called Zeus (or Guardian 4). This upcoming CGM will require only one calibration upon installation (i.e., entering a capillary blood sugar measurement into the system to ensure accuracy).
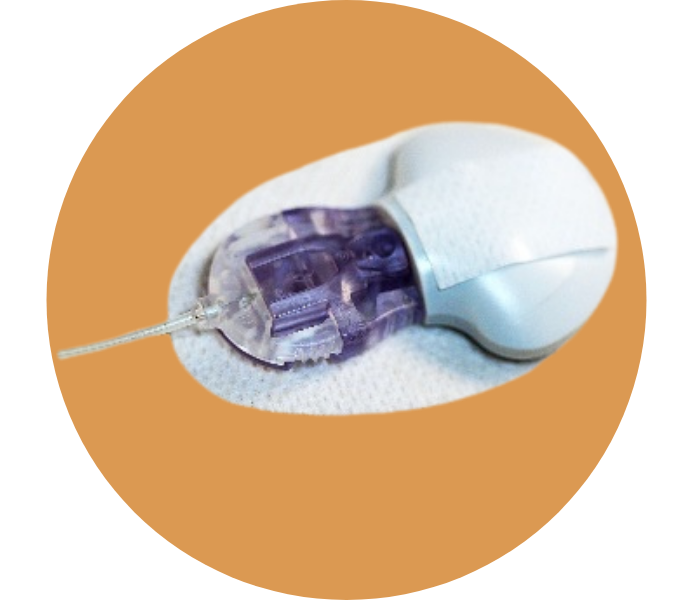
MiniMed Duo
The MiniMed Duo, currently available in Europe, is the first tool to reduce the number of devices by combining the infusion set and CGM sensor into one device that lasts three days.
At the current time, this groundbreaking technology seems to be working with one of Medtronic’s older pumps—the MiniMed Veo—, but it certainly opens the door to a future with fewer devices.
… And many more
Here’s a sneak peek at more products that are in the works:
- An insulin pump with two hormones (glucagon and insulin) that could further reduce the risk of hypoglycemia.
- Continuous ketone monitoring devices to reduce the risk of severe hyperglycemia (ketoacidosis) and manage sick days.
- A medication to delay the onset of T1D in higher-risk patients (e.g., children of a person with T1D).
- A longer-acting type of insulin that only requires one weekly injection.
- CGMs that last several months.
- Etc.
With this constantly evolving technology, it looks like the daily lives of people with T1D will keep on improving.
Now, here’s hoping that the costs will be reasonable and that people who want to use these treatments and this technology will have access to them through government insurance coverage.
References (part 1 & 2):
- Tandem 2020 ANNUAL REPORT, consulté le 23 juin 2021, https://tandemdiabetes.gcs-web.com/static-files/83769c0b-f6b2-4c3e-b050-7e246e7ebfc7
- Insulet’s Omnipod® Horizon™ Hybrid Closed-Loop System Improves Glycemic Control in Patients as Young as Two Years Old with Type 1 Diabetes, consulté le 23 juin 2021, https://investor.insulet.com/news-releases/news-release-details/insulets-omnipodr-horizontm-hybrid-closed-loop-system-improves
- Medtronic MiniMed 780G System User’s Manual, consulté le 23 juin 2021, https://www.manualslib.com/manual/1971363/Medtronic-Minimed-780g.html
- Médicaments biosimilaires, consulté le 23 juin 2021, https://www.quebec.ca/sante/systeme-et-services-de-sante/medicaments-biosimilaires
- Insulines biosimilaires – Ce que vous devez savoir, consulté 24 juin 2021, https://www.frdj.ca/insulines-biosimilaires-ce-que-vous-devez-savoir/
- Dexcom Looks to the Future of Continuous Glucose Monitoring, consulté le 24 juin 2021, https://dexcom.gcs-web.com/news-releases/news-release-details/dexcom-looks-future-continuous-glucose-monitoring
- CONNECTED CARE: Connected-Pen Approval, consulté le 24 juin 2021, https://www.lillymedical.com/en-us/answers/connected-care-connected-pen-approval-104876
- NovoPen 6 and NovoPen Echo Plus: Connected Insulin Pens To Launch in Early 2019, consulté le 24 juin 2021, https://diatribe.org/novopen-6-and-novopen-echo-plus-connected-insulin-pens-launch-early-2019
Sign up for our newsletter
Subscribe to stay informed about type 1 diabetes.
Participate in the BETTER registry!

First registry of people living with T1D in Canada.
Learn More






