The Eversence continuous glucose monitoring (CGM) system, manufactured by Senseonics/Ascencia and available in the US since 2018, has revolutionized the world of CGMs with a sensor that has been approved for 90 days (three months)—versus 7 to 14 days for other CGMs (Dexcom, Medtronic and Freestyle Libre).
An earlier version that was launched in Europe in 2017, Eversense XL, features a sensor with a longer lifespan that can be worn for 180 days (six months).
The manufacturer is currently working on enhancing this earlier version to offer a sensor that is as accurate as the one available in the US and that lasts for six months.
How does it work?
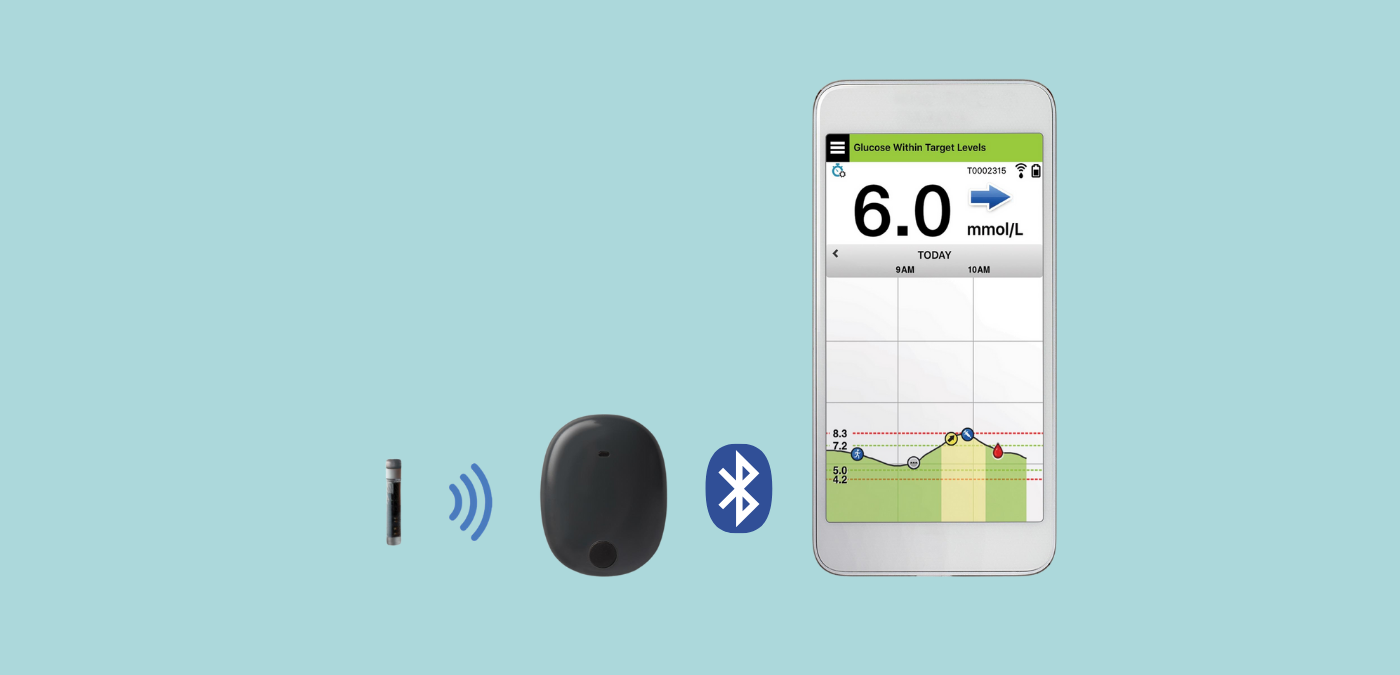
Eversense works with an under-the-skin sensor, a removable and rechargeable smart transmitter, and a smart phone app that receives blood sugar data.
The sensor is inserted under the skin of the upper arm by a certified healthcare professional.
The rechargeable transmitter, which does not need to be replaced, is then glued to the skin right above the sensor to collect the data and send it to the app.
Only one caveat: the system requires a daily calibration, i.e., entering a capillary blood sugar measurement into the system to ensure accuracy.
Not only is this system the only one that features a sensor that is inserted under the skin for a long period, but it also offers the option of a vibrating alert from the transmitter on the skin as well as a phone alert in case of low or high blood sugar.
How reliable is it?
The findings of a study conducted on the latest system and its six-month sensor were presented on June 27, 2021, at the American Diabetes Association (ADA) virtual 81st Scientific Session.
The study found that the CGM system is reliable and as accurate as other CGMs currently on the market.
The new sensor can be worn for a longer period thanks to a new chemical formula, and provides sustained accuracy across the board (first days of wear, hypoglycemia and hyperglycemia, etc.).
Also, no adverse events were reported in the study with regard to the insertion and removal of the sensor, and very few minor skin infections (0.36% in a total of 558 procedures).
So, what’s next?
The first-generation Eversense (three months) should be available in Canada in three to five years, and the wait will be even longer for the latest generation (six months). And we still don’t know whether this technology will be covered or not.
In June 2018, the manufacturer announced a partnership with Beta Bionics—a company that is currently developing an artificial pancreas—to develop a “bionic pancreas” system.
This new-generation CGM is definitely paving the way for revolutionary technology.
According to the BETTER registry (a kind of census of people with type 1 diabetes in Quebec), close to 80% of patients already use a CGM.

Want to learn more about what’s coming up in type 1 diabetes? This webinar presented by Dr. Rabasa-Lhoret should answer your questions.
Sign up (or sign your child up) for the BETTER Registry to be able to participate. Learn more »
Reference
- ADA 2021. Presented on June 27, 2021. Abstracts 149-OR
Participate in the BETTER registry!

First registry of people living with T1D in Canada.
Learn More